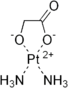You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
Pt content in Pt based pills.
- Thread starter andu
- Start date

Help Support Gold Refining Forum:
This site may earn a commission from merchant affiliate
links, including eBay, Amazon, and others.
Martijn
Well-known member
Never heard of it. do you have some info on them? are these medicinal platinum pills?
Martijn
Well-known member
I just read two of them and can't find any platinum on the ingredients. There's probably more platinum in a liter of seawater.
Health scam? or do you have a brand that actually holds value?
these are mostly vitamin pills as far as i can see.
Health scam? or do you have a brand that actually holds value?
these are mostly vitamin pills as far as i can see.
I've recycled cis and carboplatin. Nasty toxic stuff, but have done it. Is this what you refer to?
I wish i did specify, yes cis and carbo, and likes is what i had in mind, they all seem the same eventho there's so many brands ofc. No idea if i can get an actual supply but maybe i can stumble upon a few. No idea what they called over here either yet I imagine they of similar composition  .
.
Always bad news to stumble upon PGMs for this matter, however they tend to have quite a heavy presence wherever you supposed to find any.
I did sell all my PGM scrap but I wouldn't and this seems quite size-manageable so I would like to know if to hold.
Always bad news to stumble upon PGMs for this matter, however they tend to have quite a heavy presence wherever you supposed to find any.
I did sell all my PGM scrap but I wouldn't and this seems quite size-manageable so I would like to know if to hold.
I never got pills. It was all compounds. I seem to recall paying almost nothing for them. I can’t remember precisely but I think did destroy them by fuming in nitrosulfuric and then boiling in aqua regia, This was a long time ago.
So these are antineoplastic (anti-cancer) drugs, obviously. It would require a chemical process, since the platinum is covalently bound within the molecule.
Might also require different processes for the different drugs.
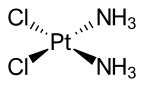
Cisplatin is Pt bound to 2 chlorines and 2 ammonia ligands.
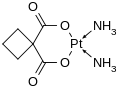
Carboplatin also has two ammonia ligands, but the other 2 binds are metalloester bonds to an organic molecule (cyclobutan-1,1-dicarboxylic acid). It's derived from cisplatin. Interestingly, in the reaction which creates it, silver nitrate is used.
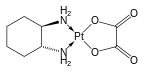
Oxaliplatin again derives from the base drug cisplatin, contains a dicarboxylic acid group, but instead of ammonia groups, it features 2 bonds to trans-1,2-diaminocyclohexane.
The key is going to be finding a reaction which will liberate the Pt from those various side groups. I suspect cisplatin might be converted to tetrachloroplatinate ion in a reaction with HCl, given cisplatin's synthesis appears to be done under basic conditions with silver nitrate and potassium chloride. HCl should cleave off the ammonia groups, creating acidic ammonium salts. It might require the presence of a chloride salt to facilite the additional ions. The tetrachloroplatinate should then be able to be directly reduced.
But the other 2 drugs could be more complicated with their organic side groups.
Might also require different processes for the different drugs.
Cisplatin is Pt bound to 2 chlorines and 2 ammonia ligands.
Carboplatin also has two ammonia ligands, but the other 2 binds are metalloester bonds to an organic molecule (cyclobutan-1,1-dicarboxylic acid). It's derived from cisplatin. Interestingly, in the reaction which creates it, silver nitrate is used.
Oxaliplatin again derives from the base drug cisplatin, contains a dicarboxylic acid group, but instead of ammonia groups, it features 2 bonds to trans-1,2-diaminocyclohexane.
The key is going to be finding a reaction which will liberate the Pt from those various side groups. I suspect cisplatin might be converted to tetrachloroplatinate ion in a reaction with HCl, given cisplatin's synthesis appears to be done under basic conditions with silver nitrate and potassium chloride. HCl should cleave off the ammonia groups, creating acidic ammonium salts. It might require the presence of a chloride salt to facilite the additional ions. The tetrachloroplatinate should then be able to be directly reduced.
But the other 2 drugs could be more complicated with their organic side groups.
- Joined
- Aug 24, 2010
- Messages
- 172
Most are chemotherapy medications, e.g. carboplatin. C6H12N2O4Pt. brand name Paraplatin. molar mass 371.249 g/mol. There also are cisplatin, nedaplatin, and oxaliplatin. Several others in clinical trials, not marketed yet. Each type will pose challenges for recovery, yet, it can be done, and yields may be allright, if you're paying little to nothing for the material. Be safe, and dispose of waste products responsibly.Never heard of it. do you have some info on them? are these medicinal platinum pills?