kurt said:
I have never processed any dental scrap so I could be wrong here but as I understand it because of the mercury used in dental work don’t you first need to put the dental scrap through a retort process to safely remove/recover the mercury before doing any melting (inquartation) &/or before doing any chemical process?
Kurt
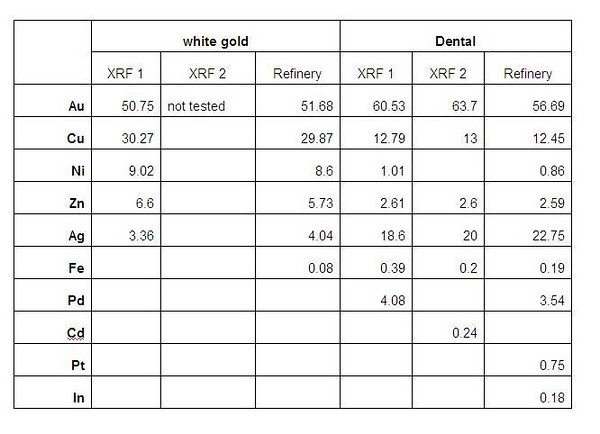
That applies only to silver amalgam, but there's more to it than just looking for fillings. It has been common practice for dentists to cap teeth that are filled. In such a case, the crown will hide the presence of mercury, but it's easy enough to uncover. I used to smash the tooth with a small hammer, which will remove the crown, and separate the filling from both the crown and tooth. The filling will have a dull gray appearance, so it's easy to sort from the broken tooth and the crown.
If the crown shatters, it's not worth processing. I encountered several that were platinum group, but each is ductile enough to withstand breaking the tooth without shattering. If there is doubt, each one may need to be tested, which can be done by dissolving a sample with a little AR. The whole cleaned crown can be tested, which will speed up the test. If the solution is dark green, pretty good chance there's no value present, but it should be tested with stannous chloride. If the solution is a dark brown color, or leans green with brown highlights, pretty good chance there's value present.
Nick offered some excellent advice in regards to processing dental waste. By inquarting, the vast majority of the platinum metals will be sorted from the gold, and will be easily recovered at a later date from the slimes of a silver cell.
With dental work, if the white material leans blue/white, and is very brittle (can't be bent), it contains no values. Again, if there's doubt, do some testing, until you are very comfortable with your decisions.
Harold